Reduce Operational Risk in EU Clinical Trials Before It Becomes Regulatory Exposure
Practical frameworks, readiness tools, and structured execution guidance for sponsors, CROs, and clinical operations teams working under the EU CTR.
EU clinical trials are becoming more complex — not because teams lack expertise, but because coordination, data, systems, and regulations increasingly intersect.
EU Clinical Trial Support exists to help clinical trial teams see clearly, prepare properly, and execute with confidence — whether a trial is about to start or already in progress.
A structured, independent support layer for EU clinical trial operations.
Where EU Clinical Trials Break Down
Clinical trials rarely fail because of science. They fail because of execution complexity — small structural weaknesses that compound across countries, vendors and regulatory layers.
80–90%
of trials experience delays during planning, activation or enrollment.
~50%
fail to meet enrollment timelines.
60%+
require protocol amendments during execution.
~30%
of Phase III failures are significantly influenced by operational issues.
Fragmented Documentation
- Inconsistent version control
- Duplicate evidence across sponsor and CRO
- Unclear ownership of key files
Cross-Country Inconsistency
- Different ethics expectations
- Variations in local implementation
- Uneven review timelines
Ethics Committee Delays
- Clarification cycles
- Administrative completeness gaps
- Underestimated harmonisation effort
Regulatory Misalignment
- Compliance defined but not operationalised
- Submission strategy not mapped to workflow
- Controls not embedded in daily practice
Vendor Miscoordination
- Overlapping responsibilities
- Fragmented audit evidence
- Diluted accountability
Data Governance Blind Spots
- Unclear data ownership
- Inconsistent audit trails
- Manual reconciliation between systems
Timeline Optimism Bias
- Underestimated approval timelines
- Overestimated site activation speed
- Amendments not built into planning
The Pattern Behind the Pattern
Most EU clinical trials do not collapse because of one major event. They drift off track through distributed ownership, inconsistent documentation, cross-border friction and assumption-based planning. By the time the issue surfaces — audit, inspection, missed milestone — recovery becomes reactive and expensive.
The EU Clinical Trial Operations Framework (2026)
A practical guide for modern EU clinical trial execution.
EU clinical trials rarely underperform because teams lack expertise. They underperform when operational complexity outpaces visibility, coordination weakens between organisations, and risks surface too late.
This framework distils recurring EU clinical trial challenges into a clear, operational reference designed to support better decisions, earlier risk detection, and more controlled execution across the full EU clinical trial lifecycle.
Inside the framework
- The EU clinical trial lifecycle — end to end
- Key operational risk areas that repeatedly impact timelines
- Readiness checkpoints for planned and active trials
- Compliance considerations across EU, US, and global trials
- Where orchestration, automation, and decision support genuinely help
- A practical “next 90 days” focus model
Included formats and tools
- Framework guide (PDF)
- Audiobook version
- Podcast-style short explanations
- Operational checklists
- Readiness and risk self-assessment (planned and active trials)
Designed for
Clinical leaders who want clarity, not theory.
Get access to The EU Clinical Trial Operations Framework (2026)
Free · Practical · Confidential

Who this is for
EU Clinical Trial Support is designed for professionals who are accountable for outcomes, not just activities.
Heads of Clinical Operations
Clinical Trial Managers & Directors
Sponsors and sponsor representatives
CRO leadership and delivery teams
Clinical systems & digital transformation owners
If you are responsible for timelines, coordination, inspection readiness, or operational clarity, this site is built for you.
The reality of EU clinical trials today
Across therapeutic areas and trial sizes, the same challenges appear — repeatedly.
Operational complexity has outpaced visibility
Clinical trials now rely on multiple vendors, systems, and stakeholders. Project plans exist, but a single operational truth rarely does. Delays are often discovered late, not early.
Data exists — insight does not
CTMS, EDC, eTMF, safety systems, and vendor tools all contain information. What is missing is coherent, decision-ready visibility across them. Teams therefore manage risk reactively instead of proactively.
Vendor and stakeholder coordination is fragile
CROs, labs, imaging providers, and technology partners are essential — but handoffs are common failure points. Accountability gaps tend to appear between organisations.
Compliance spans multiple jurisdictions
EU CTR, national competent authorities, ethics committees, and global regulatory expectations (including FDA alignment) must coexist. Inspection readiness is often treated as an event rather than a state.
Change is constant
Protocol amendments, vendor changes, and scaling from one trial to many are now the norm. Processes that worked once often fail under scale.
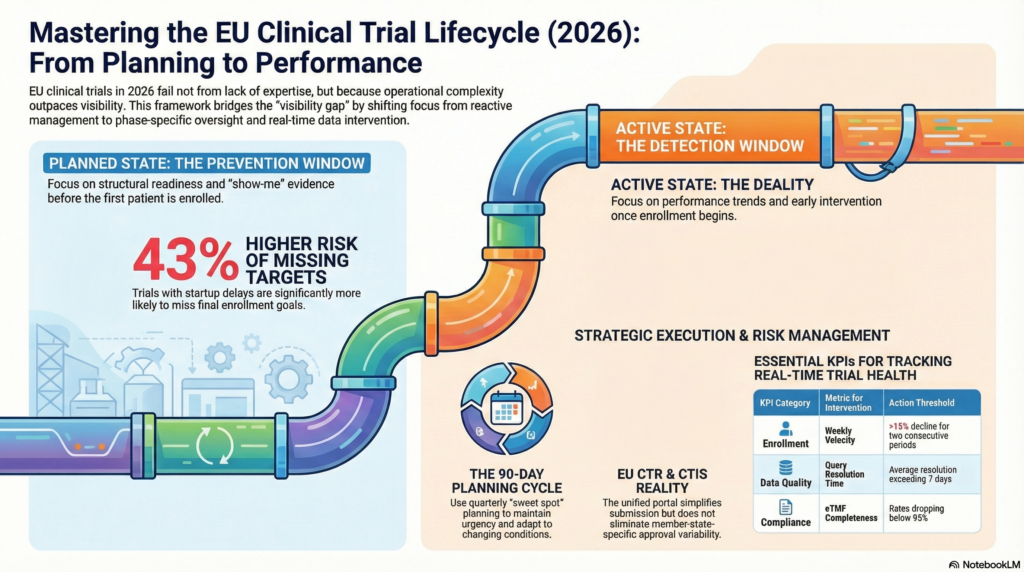
Planned vs active EU clinical trials
EU clinical trials face different risks depending on their phase. We explicitly distinguish between the two.
Planned EU clinical trials
(Not yet started)
- Underestimating startup complexity
- Poorly defined roles and handoffs
- Systems and vendors selected without orchestration in mind
- Compliance embedded too late
What matters most:
Readiness, structure, and prevention.
Active EU clinical trials
(Already running)
- Emerging bottlenecks without early warning
- Limited cross-system visibility
- Vendor coordination issues
- Growing inspection and compliance exposure
What matters most:
Early detection, correction, and controlled acceleration.
What actually helps
There is no single tool or organisation that “solves” EU clinical trials. Effective teams combine structure, visibility, coordination, and judgement. The most effective approaches typically include:
Operational diagnostics
- Trial readiness assessments
- Phase-specific risk identification
- Clear prioritisation of what matters next
Purpose: See clearly before acting.
Orchestration, not replacement
- Clear ownership and accountability
- Defined handoffs between stakeholders
- Coordination across vendors without disrupting them
Purpose: Reduce friction without replacing partners.
System landscape clarity
- Mapping CTMS, EDC, eTMF, and vendor tools
- Identifying overlaps, gaps, and blind spots
Purpose: Visibility without system replacement.
Compliance by design
- EU CTR considerations embedded into operations
- Alignment with US and global expectations
- Inspection readiness treated as continuous
Purpose: Avoid last-minute compliance stress.
Targeted automation and decision support
- Automation applied only where repetition exists
- Decision support used where complexity is high
- Human control retained at all times
Purpose: Reduce noise, not responsibility.
How EU Clinical Trial Support helps
EU Clinical Trial Support provides independent, structured support for EU clinical trial teams. Support typically includes:
- Readiness and risk assessments
- Operational and coordination diagnostics
- System and data landscape clarity
- Compliance-aware operational guidance
We remain:
- System-agnostic
- Confidential
Some teams use our frameworks internally.
Others ask us to review their situation against them.
Podcast: The EU Clinical Trial Operations Framework
The EU Clinical Trial Operations Framework (2026)
A practical guide for modern EU clinical trial execution.
EU clinical trials rarely underperform because teams lack expertise. They underperform when operational complexity outpaces visibility, coordination weakens between organisations, and risks surface too late.
This framework distils recurring EU clinical trial challenges into a clear, operational reference designed to support better decisions, earlier risk detection, and more controlled execution across the full EU clinical trial lifecycle.
Inside the framework
- The EU clinical trial lifecycle — end to end
- Key operational risk areas that repeatedly impact timelines
- Readiness checkpoints for planned and active trials
- Compliance considerations across EU, US, and global trials
- Where orchestration, automation, and decision support genuinely help
- A practical “next 90 days” focus model
Included formats and tools
- Framework guide (PDF)
- Audiobook version
- Podcast-style short explanations
- Operational checklists
- Readiness and risk self-assessment (planned and active trials)
Designed for
Clinical leaders who want clarity, not theory.
Download The EU Clinical Trial Operations Framework (2026)
Free · Practical · Confidential


EU Clinical Trial Operations Framework (2026)
Written for operational realities
What you’ll find inside
Why this book is different
It focuses on operational patterns — not generic compliance checklists — so you can recognize velocity decline, query backlog escalation, and eTMF degradation before they become crises.
You get decision frameworks with specific thresholds, plus real-world cases that show what actually happened and how teams recovered from performance and readiness breakdowns.
It also includes copy-paste tools: checklists, templates, KPI structures, troubleshooting guides, and risk assessment assets ready to adapt.
