3 EU Clinical Trial Pilot Projects now open & free
EUclinicaltrial.support is opening three pilot projects for Medtora — a compliant clinical trial platform designed to be easy to use, intuitive, and simple, without sacrificing security and regulatory discipline.
If your team is tired of tool sprawl, fragmented documentation, and slow handoffs between Sponsor, CRO, and sites, a pilot is the fastest way to validate whether a unified approach reduces friction in real operations.
Pilot places are limited to keep onboarding and support high-touch and fast.

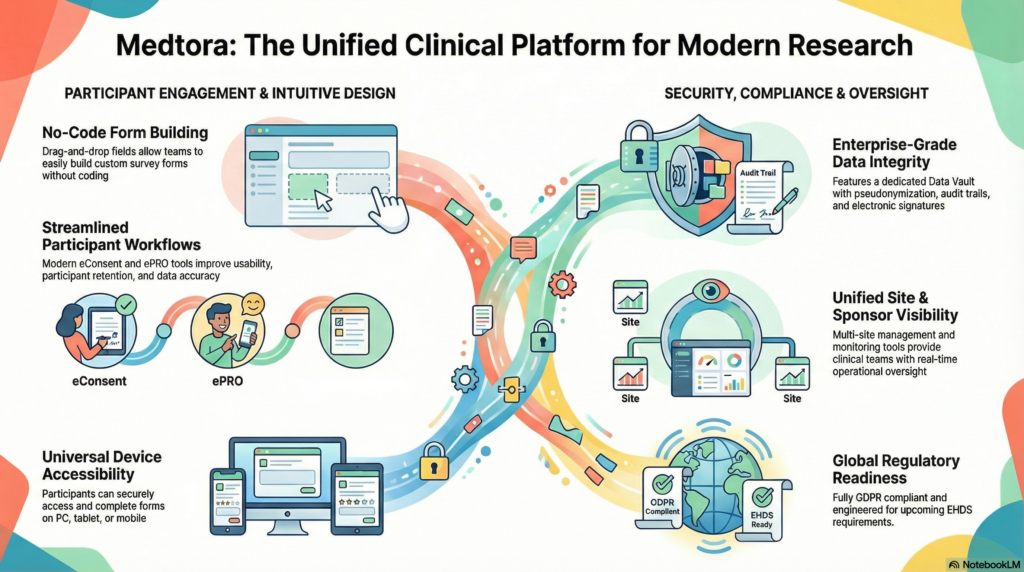
What Medtora is (in plain terms)
A unified platform bringing security, compliance posture, and cross-organizational collaboration into one clinical environment — with access management, consent/compliance workflow support, and document/media handling in one place.
- Unified access & collaboration: controlled roles and permissions across stakeholders.
- Consent & compliance support: structured handling of compliance-relevant workflows and artifacts.
- Documents & evidence: reduce fragmentation by keeping operational evidence together.
- Security positioning: built around secure access and controlled authentication.
Features converted to buyer benefits
Unified access & collaboration
Benefit: fewer handoffs, fewer “who owns this?” moments, clearer responsibility.
- Controlled roles/permissions across internal and external users.
Consent & compliance built-in
Benefit: reduces consent chaos and audit anxiety with structured, traceable artifacts.
- Consent and compliance workflows supported within the platform.
Documents, media & evidence in one place
Benefit: less fragmentation, fewer duplicates, faster inspection readiness confidence.
- Document/media handling designed for clinical project use.
Security & authentication discipline
Benefit: less exposure from ad-hoc sharing and unmanaged access.
- Secure access patterns and controlled authentication.
Data protection & retention structure
Benefit: fewer governance blind spots by treating protection/retention as operational requirements.
- Designed to support strong data governance expectations.
Pilot program (3 projects)
Who should apply
- Sponsor or CRO running an EU study with cross-organization collaboration needs
- Teams that want fewer systems and clearer evidence structure
- Organizations that want a compliant posture without operational complexity
What you get in the pilot
- Fast onboarding and configuration for a real project
- Implementation support for roles, consent/compliance workflows, and document handling
- Feedback loop that directly shapes the roadmap
- Clear success criteria (time saved, reduced fragmentation, fewer handoffs)
Pilot success criteria (what we measure)
- Reduction in duplicate document handling
- Faster role onboarding/offboarding
- Fewer “where is the latest version?” incidents
- Improved inspection readiness confidence
- Shorter cycles for participant and consent administration tasks
- Less system sprawl (fewer tools needed per study)
Buyer checklist (tick off) — with Medtora answers
Use this to evaluate any clinical platform. Tick what you require. The right column shows how Medtora addresses it (or validates it in the pilot).
Yes (core design)
Unified collaboration with controlled access; validated in the pilot with your real stakeholder map.
Yes
Access and roles are central; pilot confirms speed and clarity in real onboarding.
Pilot validates
Designed to reduce fragmentation; we measure how many external trackers you can retire.
Yes
Document/media handling is designed to reduce fragmentation; pilot confirms your inspection-readiness workflow.
Pilot validates
We configure the structure with you and measure reduction in duplicates and version confusion.
Yes (security posture)
Designed around controlled access; pilot verifies it matches your governance requirements.
Yes
Consent & compliance support is included; pilot focuses on whether it simplifies real operations.
Yes (governance posture)
Built with strong data governance expectations; pilot aligns configuration to your SOPs.
Benchmark: Medtora vs common alternatives
Category-based view focused on operational reality (not marketing claims).
| Option | Typical strengths | Typical trade-offs |
|---|---|---|
| Enterprise suites CTMS/EDC/large platform stacks |
Very broad feature coverage; mature enterprise governance. | Can be heavy to implement; more complexity for teams and sites; higher change-control overhead. |
| Point solutions Multiple tools stitched together |
Depth in one domain (eConsent, ePRO, EDC, etc.). | Tool sprawl; more logins; more handoffs; more fragmentation in evidence and workflows. |
| Mid-market platforms | Often easier than enterprise suites; clearer packaging and faster rollout. | May still require add-ons or integrations depending on your operating model. |
| Medtora pilot | Unified, security + compliance posture + collaboration approach designed to reduce daily friction. | Pilot scope limited (3 projects) to keep onboarding fast and support high-touch; not positioned as an “everything suite” on day one. |
Frequently asked questions
Why only three pilot projects?
Because the goal is speed and depth. A pilot only works if onboarding, configuration, and feedback are handled tightly and quickly—not diluted across dozens of projects.
What makes this pilot worth doing?
You get a controlled way to test whether a unified platform reduces fragmentation (documents, access, consent/compliance workflows) and improves day-to-day usability—without committing to a long rollout first.
What do you need from us to start?
A real project use-case, your stakeholder map (Sponsor/CRO/sites), and the minimum workflow you want to validate first (access roles, consent/compliance, or evidence/document handling).
Learn more about the pilot project
If you want a platform that is compliant but actually usable day-to-day, apply for one of the three pilot projects.