You Have the Framework.
Now Execute It.
The EU Clinical Trial Operations Framework (2026) gives you clarity across the lifecycle. The next step is turning the theory into controlled execution — inside a system built for EU-regulated reality.
Reduce Risk. Strengthen Compliance. Focus on the Science.
BOOK: EU Clinical Trial Operations Framework
Powered By EmbedPress
Put the Framework Into Practice With a System Pilot
The System Pilot is designed for teams that want to test operational execution in a structured environment — using a real project, with clear evaluation criteria and controlled onboarding.
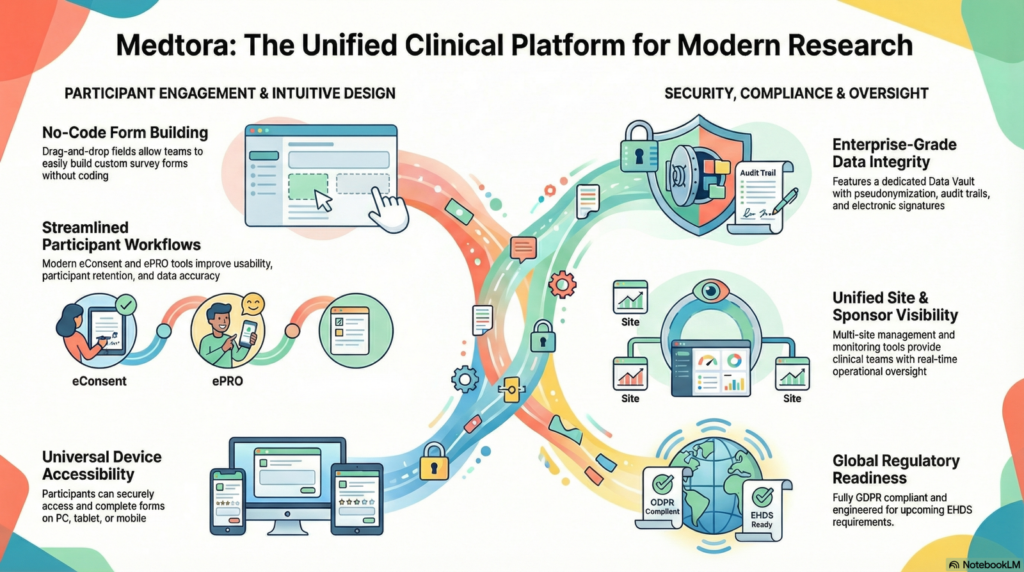
The EU CTR Reality — In One View
The infographic below is designed to help you quickly communicate what the framework covers: the lifecycle, the recurring risk areas, and where teams lose control when execution becomes multi-country and multi-vendor.
Podcast: Operational Execution in the EU
The podcast is built for trial operators who want fewer surprises: what breaks, why it breaks, and what “control” actually looks like in EU-regulated execution.
Listen with your team and use the ideas to pressure-test your workflow—then decide if a structured pilot makes sense.
- Execution signals teams miss until pressure hits
- EU CTR friction points that create hidden delays
- Implementation patterns that reduce rework
